Open Response Question 1
The values of the first three ionization energies (I1, I2, I3) for magnesium and argon [in kJ/mole] are as follows:
Mg --> I1: 735
I2: 1443
I3: 7730
Ar --> I1: 1525
I2: 2665
I3: 3945
(a) Give the electronic configuration of Mg and Ar.
(b) In terms of these configurations, explain why the values of the first and second ionization energies of Mg are significantly lower than the values for Ar, whereas the third ionization energy of Mg is much larger than the third ionization energy Ar.
(c) If a sample of Ar in one container and a sample of Mg in another container are each heated and chlorine is passed in to each container, what compounds, if any , will be formed? Explain in terms of the electronic configuration given in part (a).
(d) Element Q has the following first three ionization energies [in kJ/mole]:
I1: 496
I2: 4568
I3: 6920
What is the formula for the most likely compound of element Q with chlorine? Explain the choice of formula on the basis of the ionization energies.
Mg --> I1: 735
I2: 1443
I3: 7730
Ar --> I1: 1525
I2: 2665
I3: 3945
(a) Give the electronic configuration of Mg and Ar.
(b) In terms of these configurations, explain why the values of the first and second ionization energies of Mg are significantly lower than the values for Ar, whereas the third ionization energy of Mg is much larger than the third ionization energy Ar.
(c) If a sample of Ar in one container and a sample of Mg in another container are each heated and chlorine is passed in to each container, what compounds, if any , will be formed? Explain in terms of the electronic configuration given in part (a).
(d) Element Q has the following first three ionization energies [in kJ/mole]:
I1: 496
I2: 4568
I3: 6920
What is the formula for the most likely compound of element Q with chlorine? Explain the choice of formula on the basis of the ionization energies.
Open Response Question 2
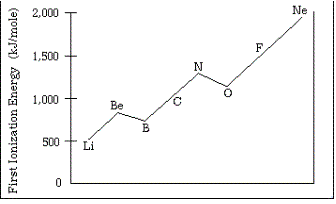
The above diagram shows the first ionization energies for the elements from Li to Ne. Briefly explain each of the following statements.
(a) In general, there is an increase in the first ionization energy from Li to Ne.
(b) The first ionization energy of B is lower than that of Be.
(c) The first ionization energy of O is lower than that of N.
(d) Predict how the first ionization energy of Na compares to those of Li and of Ne. Explain.
(a) In general, there is an increase in the first ionization energy from Li to Ne.
(b) The first ionization energy of B is lower than that of Be.
(c) The first ionization energy of O is lower than that of N.
(d) Predict how the first ionization energy of Na compares to those of Li and of Ne. Explain.
Multiple Choice Questions
In THIS practice exam, answer questions 30, 31, 33-41