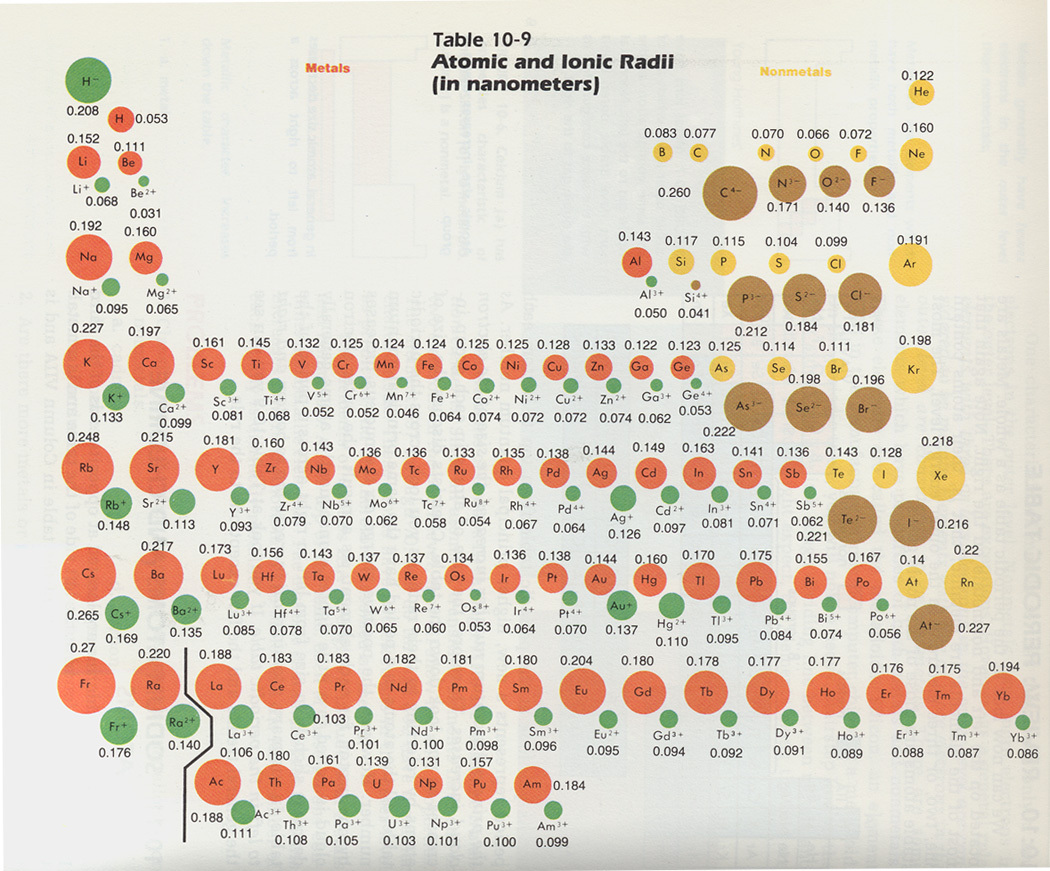
Atomic Radii/Ionic Radii
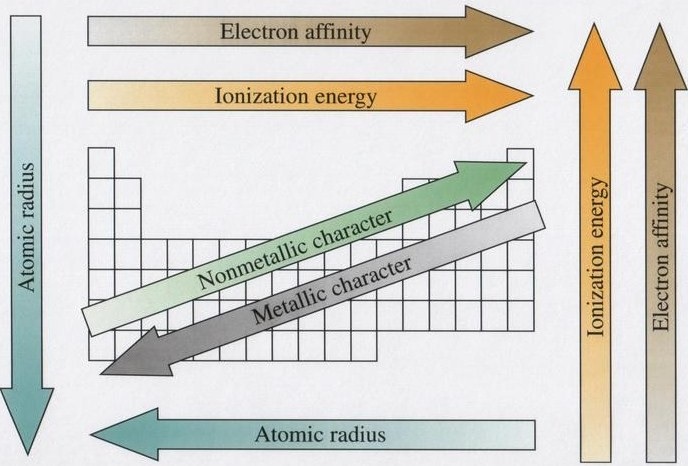
Down a group, atomic radius increases because the number of electrons increases, resulting in a larger electron cloud. It also will have more electron shells, and the farther out an electron shell is the less subject those electrons are to the pull of the nucleus, and the farther away from the nucleus they will travel.
Across a period, atomic radius decreases because the increasing number of protons results in a higher "effective nuclear charge" (ability of the nucleus to pull electrons towards itself); the electrons being added to each orbital are all subject to this higher effective nuclear charge. Ionic radii depend on whether an atom forms a positive or negative ion (covered later). When an atom forms a negative ion, it becomes larger because the addition of an electron causes repulsion within the electron cloud, causing it to expand. When an atom forms a positive ion, it becomes smaller because the loss of an electron decreases repulsion within the electron cloud - and often will cause the atom to lose an entire energy level, significantly reducing the size of the atom.
Across a period, atomic radius decreases because the increasing number of protons results in a higher "effective nuclear charge" (ability of the nucleus to pull electrons towards itself); the electrons being added to each orbital are all subject to this higher effective nuclear charge. Ionic radii depend on whether an atom forms a positive or negative ion (covered later). When an atom forms a negative ion, it becomes larger because the addition of an electron causes repulsion within the electron cloud, causing it to expand. When an atom forms a positive ion, it becomes smaller because the loss of an electron decreases repulsion within the electron cloud - and often will cause the atom to lose an entire energy level, significantly reducing the size of the atom.
Ionization EnergyIonization Energy: the minimum amount of energy required to remove an electron from an atom
First ionization energy increases across a period to the right and up a group (within a group, the lower the atomic number, the higher the ionization energy). Subsequent ionization energies must then be deduced based on the size of the atom's nucleus & the energy levels/configuration of its remaining electrons. This is because, across a period and up a group, the atomic radius decreases, meaning the outermost electrons are closer to the nucleus & are subject to stronger nuclear attractions, requiring more energy to be pulled away. |
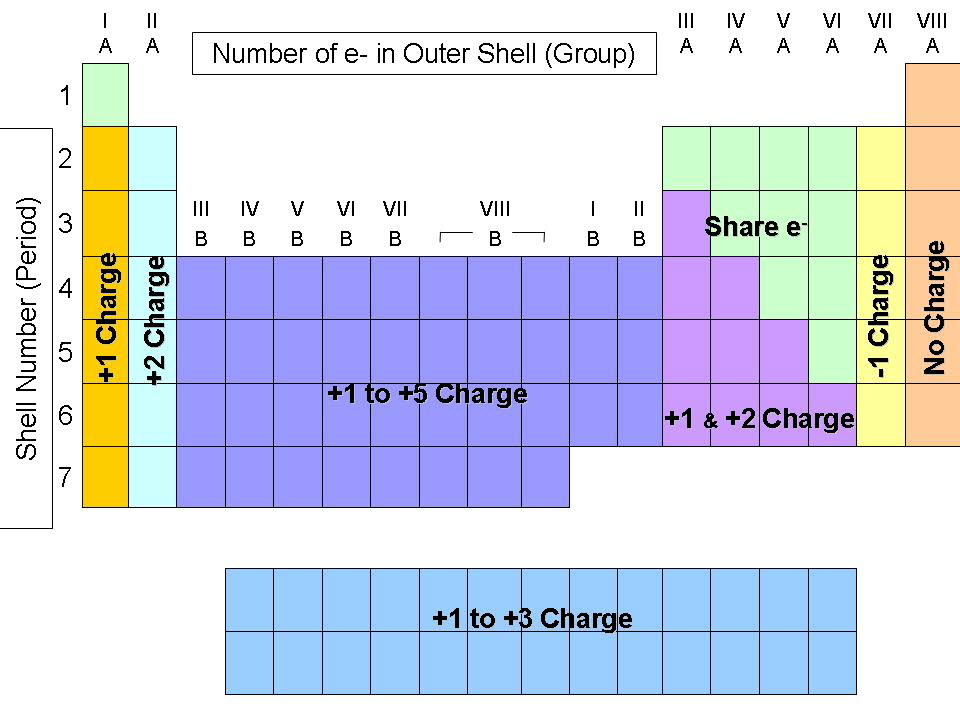
Ionic Charge
|
The charge of the ions typically formed by an atom depend largely on its electron configuration and the number of electrons in its valence shell. For example, alkali-earth metals (group 2) will give up two electrons (gaining a +2 charge) because its valence shell has only two electrons. It is thus much easier to give them up and achieve a stable octet than to gain six electrons. On the other hand, halides (second group from the right), will gain an electron because they are only one electron short of a full, stable octet - it is thus easier for them to gain one than to lose the 5 in their final p sublevel.
|
Reactivity
Reactivity trends are different for metals and nonmetals.
METALS:
Reactivity increases as you go down in a group, since the effective nuclear charge on the outermost electrons lessens significantly, which makes them much easier to strip away.
Reactivity decreases as you go across a period, since the greater number of valence electrons results in a more cumbersome process for getting rid of them. The larger nuclei across a period also increases pull on valence electrons, making them more difficult to shed.
NONMETALS:
Reactivity increases as you go up in a group, since the effective nuclear charge is greater - increasing electronegativity/electron affinity, and causing these elements to more readily take up electrons.
Reactivity increases as you go across a period, since the greater number of valence electrons makes it easier to attain a full, stable octet of electrons. The larger nuclei across the period also increases electron affinity.
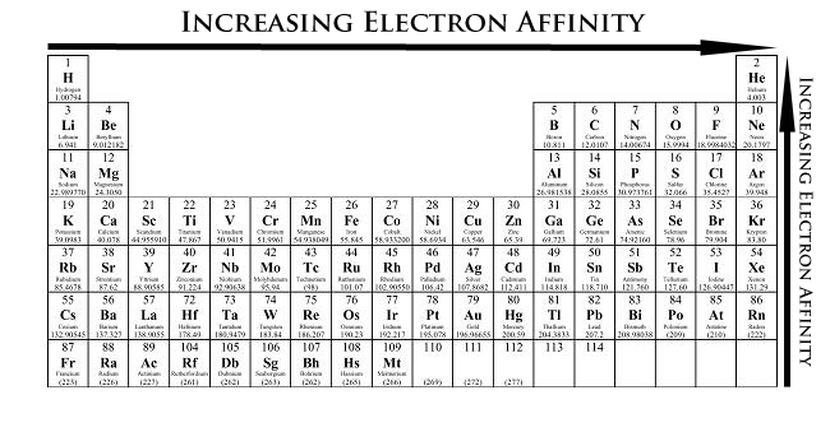
The image below shows the general trend of electron affinity (just ignore the noble gases), which ties in with reactivity in that the elements at the high and low extremes of electron affinity (like sodium and fluorine) are the most reactive.
METALS:
Reactivity increases as you go down in a group, since the effective nuclear charge on the outermost electrons lessens significantly, which makes them much easier to strip away.
Reactivity decreases as you go across a period, since the greater number of valence electrons results in a more cumbersome process for getting rid of them. The larger nuclei across a period also increases pull on valence electrons, making them more difficult to shed.
NONMETALS:
Reactivity increases as you go up in a group, since the effective nuclear charge is greater - increasing electronegativity/electron affinity, and causing these elements to more readily take up electrons.
Reactivity increases as you go across a period, since the greater number of valence electrons makes it easier to attain a full, stable octet of electrons. The larger nuclei across the period also increases electron affinity.
The image below shows the general trend of electron affinity (just ignore the noble gases), which ties in with reactivity in that the elements at the high and low extremes of electron affinity (like sodium and fluorine) are the most reactive.
These two videos explain in greater depth the kinds of trends exhibited in the periodic table, and why those trends exist.
NOTE: about the first half of this video is a review of determining electron configurations. Since that will be covered in the Atomic Structure review, you can skip to the part where it is explained how you can figure out trends in reactivity based on the valence shell configurations of different groups of the periodic table.